
Department of Chemistry and Biochemistry
Associate Professor
Research Areas: Organic
Office: 362 Chemistry Building
Lab: 331 Chemistry Building
Phone: (334) 844-6910
Fax: (334) 844- 6959
Email: blm0022@auburn.edu
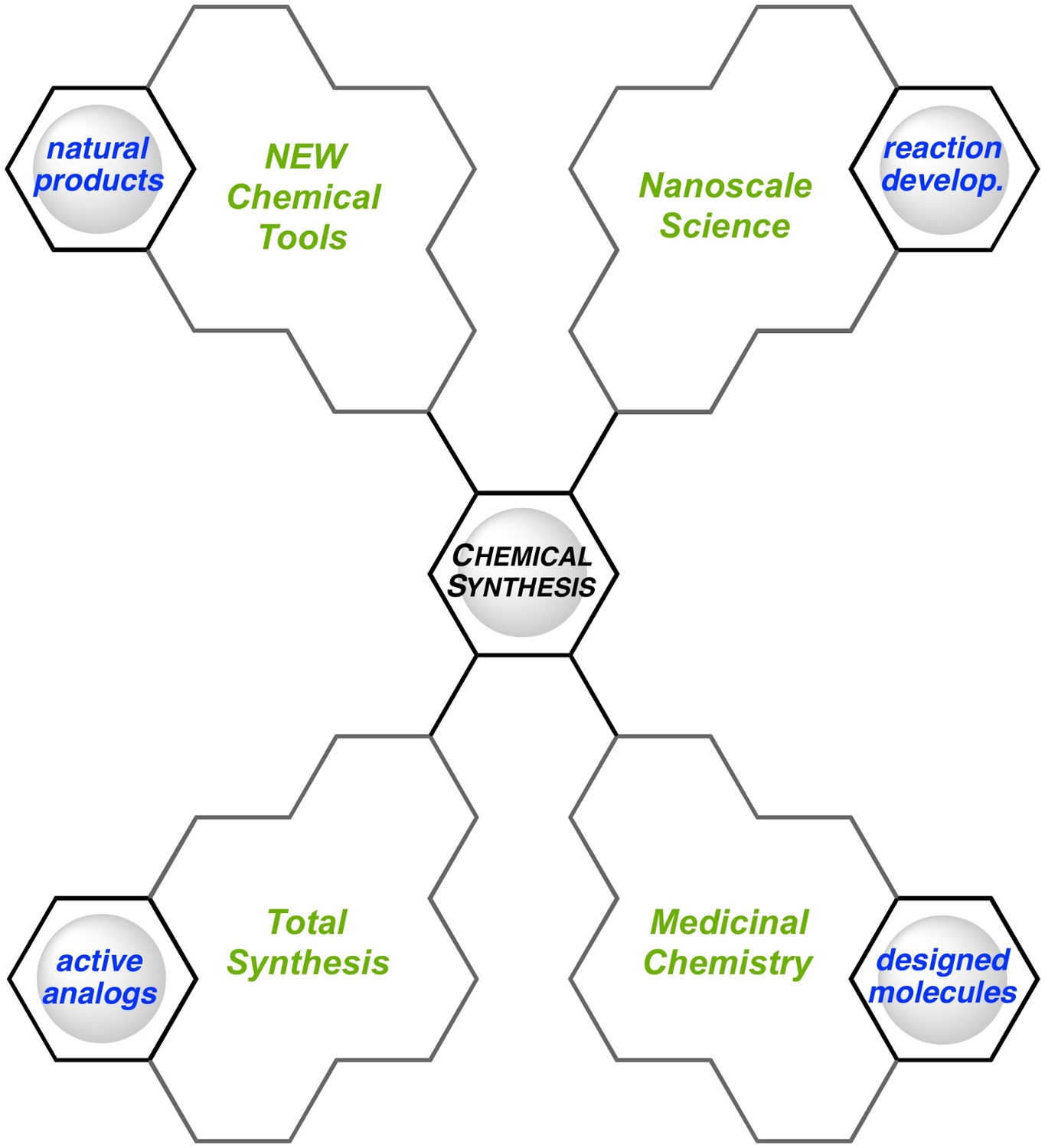
The central theme of our group’s research program is target-oriented chemical synthesis (vide infra). Under this vastly defined area, our group will concentrate on the synthesis of architecturally complex molecules that are relevant to medicinal chemistry and nanoscale science. We are interested in the synthesis of natural products, analogues thereof, nucleic acid modifications that can be used as gene silencing therapeutics, and curved aromatic systems that will serve as molecular templates in the bottom-up chemical synthesis of carbon nanotubes (CNTs). Deeply rooted in all of the synthesis projects carried out in our laboratories will be the development of new synthetic technologies that address current weaknesses that exist in the arena of chemical synthesis. It is hoped that this vision will facilitate the discovery of new, powerful synthetic disconnections that can ultimately be used to streamline synthetic approaches to bioactive compounds, enable analogue synthesis of more potent medicines, and improve the current state of the art for CNT synthesis.
Graduate students interested in joining the group should apply directly to graduate program at Auburn University within the Department of Chemistry and Biochemistry. Interested undergraduate students should send an E-mail to blm0022@auburn.edu or stop by my office to arrange a meeting.
All interested postdoctoral applications should include a current CV, copies of relevant publications, a concise summary of research experience (up to 5 pages), and at least two letters of recommendation sent under a separate cover. These can be sent via E-mail to: blm0022@auburn.edu
Mitra, N. K.; Corzo, H. H.; Merner, B. L. A Macrocyclic 1, 4-Diketone Enables the Synthesis of a p-Phenylene Ring that Is More Strained than [4] Cycloparaphenylene. Org. Lett. 2016, 18, 3278-3281.
Mitra, N. K.; Meudom, R.; Corzo, H. H.; Gorden, J. D.; Merner, B. L. Overcoming Strain-Induced Rearrangement Reactions: A Mild Dehydrative Aromatization Protocol for the Synthesis of Highly Distorted p-Phenylenes. J. Am. Chem. Soc. 2016, 138, 3235-3240
Mitra, N. K.; Meudom, R.; Gorden, J. D.; Merner, B. L. A Non-Cross-Coupling Approach to Arene-Bridged Macrocycles: Synthesis, Structure and Direct, Regioselective Functionalization of a Cycloparaphenylene Fragment. Org. Lett. 2015, 17, 2700-2703.
- Selected as an ACS Editor’s Choice Article (May 8, 2015)
- One of 10 most accessed articles for May 2015
Last updated: 11/15/2024
