Summer Bridge Students Showcase Research at Closing Ceremony
Incoming freshmen planning to study science, technology, engineering or math (STEM) recently took part in the STEM Summer Bridge Program on Auburn’s campus. Hosted by the Auburn University College of Sciences and Mathematics (COSAM) and the Samuel Ginn College of Engineering, the students prepared for college life from June 2-29. One major component of the program was preparing the students for research.

COSAM's Program Coordinator Julian Oliver and Assistant Dean for Inclusion, Equity and Diversity Dr. Kimberly Mulligan-Guy mentored students in this intensive program.
The participants were placed in interdisciplinary groups consisting of students from COSAM and the College of Engineering, where they conducted research related to the topic of water quality. The results were presented during the Summer Bridge closing ceremony on June 29.
Asha-Dee N. Celestine, assistant professor in the Department of Aerospace Engineering, and Holly Ellis, William P. Molette professor in the Department of Chemistry and Biochemistry, served as faculty advisors for the research projects. They each said they feel that research experience for incoming freshmen is important because it further develops critical thinking skills, such as analysis, problem-solving and communication.
“Working in groups also helps our students cultivate effective teamwork skills,” Celestine added.
Below is a summary of the students’ presentations.
Water Purification through Distillation
By Bryce Ellis, Kimberly Harrison, Jorden Shelton, Alex Stanley, Deven Rivera, Tony Pereira
The students presented the idea that cleaner water could help prevent 9.1 percent of global disease and 6.3 percent of all deaths. Their research focused on an experiment which aimed to use affordable and accessible filtration systems to bring clean water to places that previously did not have it.
Water samples from Lake Chewacla were collected and three homemade filtration systems were created to purify water: activated carbon, distillation and solar. Each sample of filtered water was tested for various contaminants using test strips. The results showed that distilling water can purify water from physical contaminants to achieve an EPA approved standard.
“We tried to make solutions for people out there in the world who have unfiltered water so we can give them drinkable water so we could possibly build a bridge between a water crisis and a solution, and also the most efficient way,” Bryce Ellis said of the group’s research.
The Relationship between Price and Water Quality
By Justin Cook, Kenji Guermonprez, Nadia Lane, Houston Norris, Deon Wise, Madison Wright
The students presented the fact that 2 billion people in the world use water contaminated with E. coli and that many water companies claim to have the purest water. They tested different brands of distilled water that included Voss, Fiji, Smart Water, Dasari, Sam’s Club and R.B. Water.
“We chose to buy several brands of water from most expensive to least expensive,” student Kenji Guermonprez said of the research project.
They planned to show that as price increases for water, water is purer and to show that pure water is not amenable to bacterial growth.
The study showed that as the price of consumer brand water increases, the purity of the water fluctuates along with it. Fiji water is a good example of this. All of the samples maintained the same growth of bacteria except Fiji water. It was the most expensive brand and had the most conductivity of all the sampled waters. It proved to be the harshest environment for bacteria growth.
The Comparison of Chemical Levels in High Interaction Water Sources and Low Interaction Water Sources
By Austin Darnell, Gabe Robinson, Kary Utomi, Nick Welcher, Shaniya Holley, Tia Williams, Trinitee Hughley
For this research project, the students compared water samples to test for levels of chemicals. They tested water in which there was human interaction, such as Red Barn Lake and Chewacla State Park, and low interaction, such as the pond at the Jule Collins Smith Museum. Chemical infused water was also tested. The goal was to determine if water with human interaction has higher levels of chemicals such as copper and fluoride, which causes an increase in pH levels.
“From the results, we found that the three lakes pretty much all had the same amount of chemicals in it, regardless of what interaction they had,” explained student Kary Utomi.
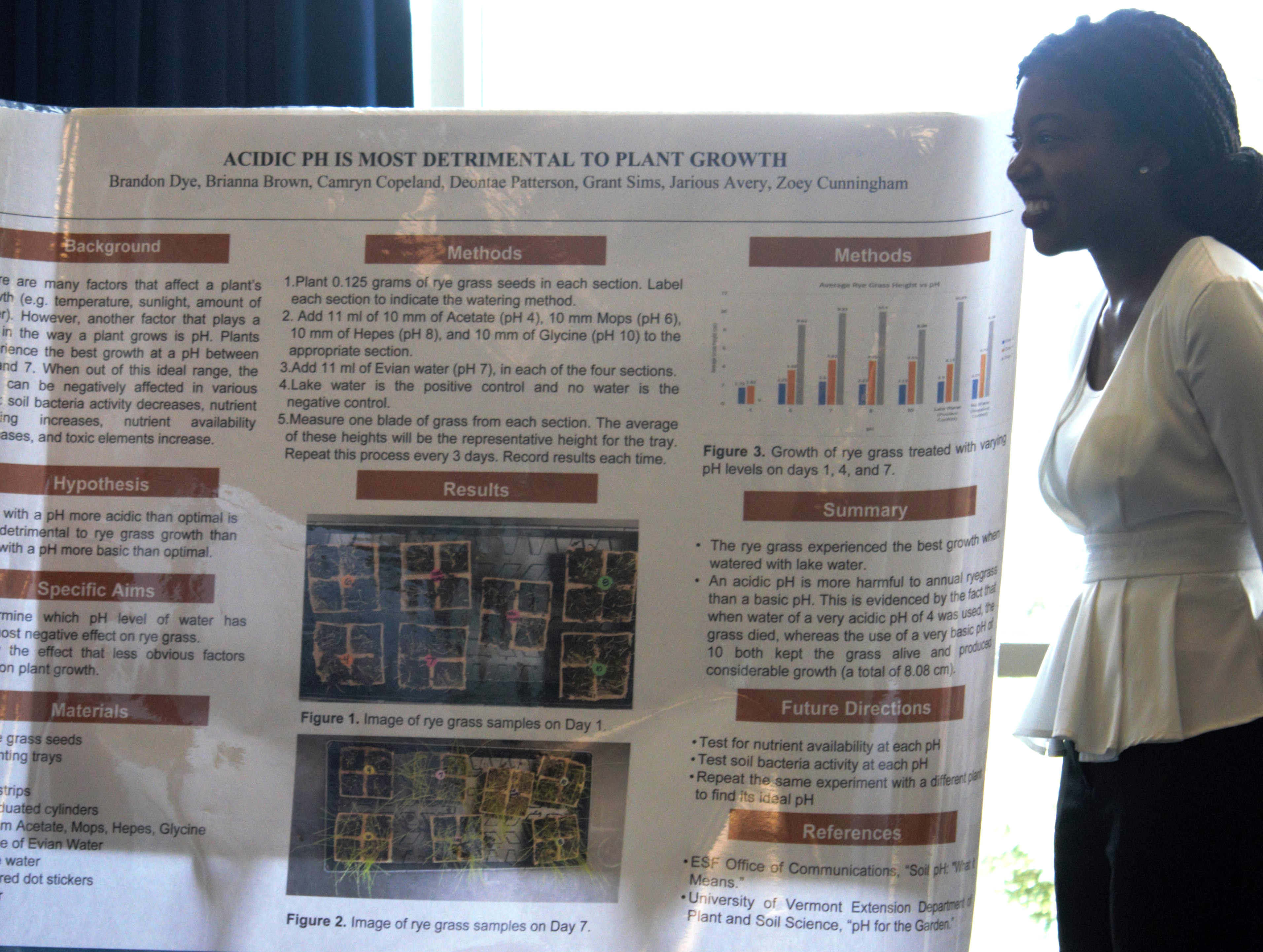
Acidic PH is Most Detrimental to Plant Growth
By Brandon Dye, Brianna Brown, Camryn Copeland, Deontae Patterson, Grant Sims, Jarious Avery, Zoey Cunningham
The group’s hypotheses explained that water with pH more acidic than optimal is more detrimental to rye grass growth than water with pH more basic than optimal. They aimed to determine which pH level of water has the most negative effect on rye grass and to show the effect that less obvious factors have on plant growth.
The students planted 0.125 grams of rye grass seeds in several sections and labeled each to indicate the watering method. They added Acetate, Mops, Hepes and Glycine and watered the grass with Evian water and lake water and measured the results every three days. The group discovered that the rye grass experienced the best growth when watered with lake water. An acidic pH is more harmful to annual ryegrass than a basic pH.
“We took multiple chemicals and we changed the pHs of our solutes to test the overall plant height progress and our results ended up being that optimal pH was seven or in that range for lake water and we had the most growth with those pHs and that the pH at four was very detrimental to the plant growth,” explained student Zoey Cunningham. “What we wanted to show was that plant life is very important for all impacts of global life.”
The Pricing of the Dorm Does Not Depend on the Quality of its Water
By Dale Feng, Kendi Davis, Elijah Kagwisa, Cara Shade, Jordan Logan, Joshua Stanton
This group of students set out to prove that the quality of water in the residence hall increases as the price of living in the residence hall increases. They planned to determine if there is a higher presence of contaminants in the more economically priced dorms on Auburn’s campus. They collected water from sinks in resident halls including South Donahue, The Village, The Hill, Cambridge and The Quad. The results showed that the price had no effect on the quality of dorms.
“We tested for lead, copper, pH, chlorine, and sulfate,” student Jordan Logan explained. “We used test strips that tested for all of these. As you can see, all the dorms all meet EPA standards. The reason that we said the higher costing dorms would have better water quality than the lower costing dorms was because age is also associated with the price. So, the older the building the less it is, so we thought there would be more contaminates under the pipes running under the older buildings, but actually the water quality is just the same.”
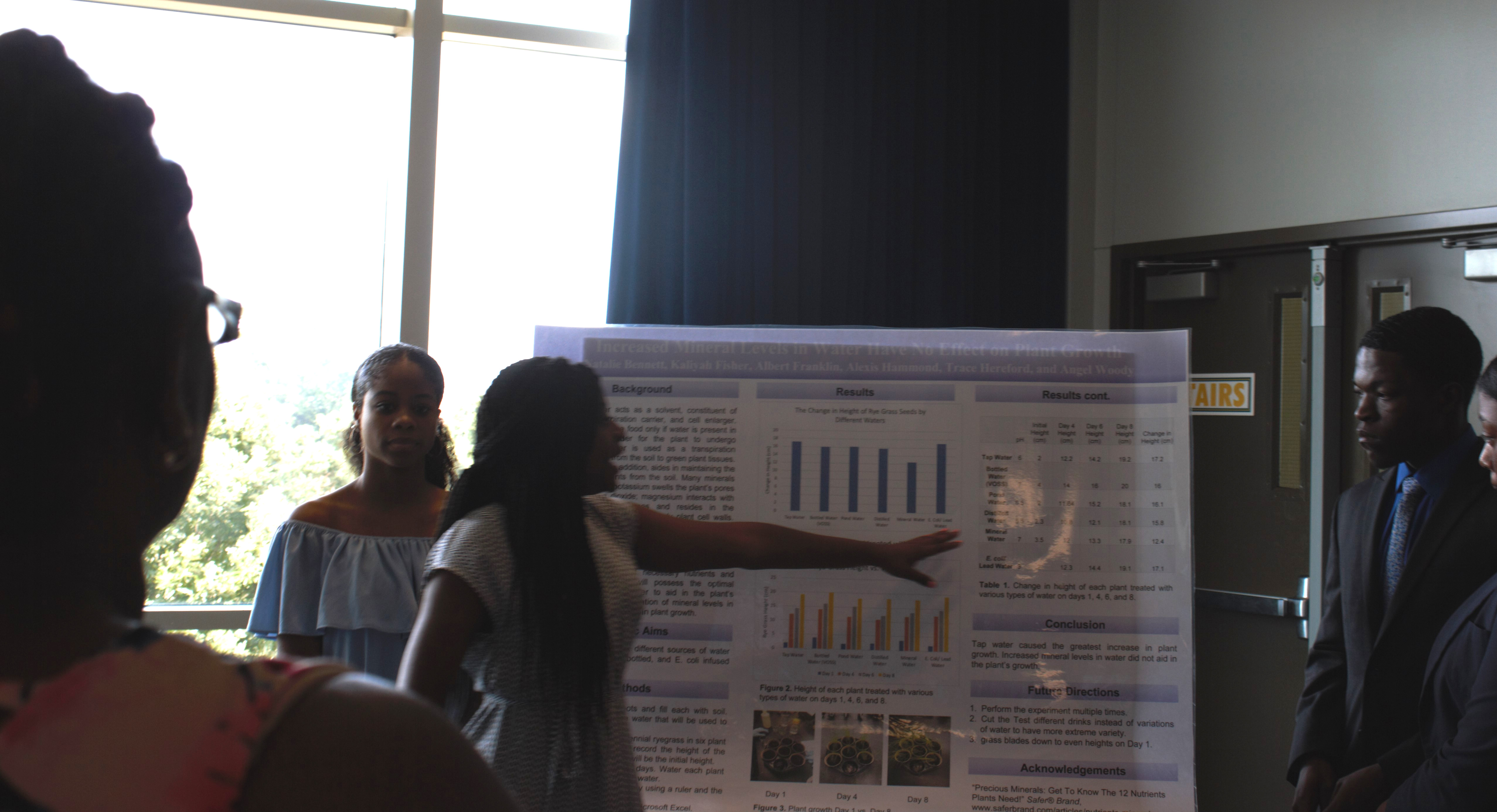
Increased Mineral Levels in Water Have No Effect on Plant Growth
By Natalie Bennett, Kaliyah Fisher, Albert Franklin, Alexis Hammond, Trace Hereford, Angel Woody
The students set out to prove that since plants thrive off of necessary nutrients and minerals, mineral water will possess the optimal amount of minerals in order to aid in the plant’s survival. Increased concentration of mineral levels in water will lead to an increase in plant growth. They planned to determine the effect of different sources of water including lakes, distilled, bottled, and E. coli infused water on plant growth. The group tested six potted plants, treated with various types of water.
“We looked up the different minerals that affect plant growth specifically, so we found potassium, magnesium, and calcium,” explained student Alexis Hammond.
“Our hypothesis was increase concentration of mineral levels of water will lead to an increase of plant growth. Our specific aim was to determine the different effects the waters that we tested; tap, bottled, lake water, E. coli infused and our man-made water,” Kaliyah Fisher added.
The group concluded that tap water caused the greatest increase in plant growth. Increased mineral levels in water did not aid in the plant’s growth.
Standing Water is Just as Contaminated as Flowing Water
By Alisha Plant, Khalil Jackson, Anthony Turner, Nolen Calvin, Su-Min Cho, Alexander Grady
This group of six students set out to prove that standing water has more contaminants than flowing water and determine what those contaminants are.
They tested the Chewacla State Park Lake and waterfall, extra distilled water and spiked water for total alkalinity, hardness, iron, copper, lead, fluoride, mercury and more.
The results proved that standing water tended to be more alkaline, have increased hardness, and a higher level of conductivity than flowing water.
Overall, the standing and flowing water had similar contaminants.
“We thought that next time we could use more samples, maybe more that weren’t necessarily centered in Auburn,” said student Anthony Turner. “We would like widen the spectrum if we had more time and more resources.”
Solar Still Substance Levels Comparable to Boiling Water Distillation
By Brandan Belser, Jr., Jazmyn Boiteux, Isaiah Davis, Jonathan Goforth, Adetola Koiki, Stephen Robinson
This group of students hoped to find a cost effective and portable method to purify water without the use of a main fuel source. They planned to prove that lake water that is purified using the solar still water process is comparable by FDA standards to the basic stovetop boiling method.
A portion of lake water was boiled on a hot plate while the other portion was placed in a solar still process to create distilled water. Test strips were placed in both samples of distilled water, both were compared based on the copper, iron, total hardness and fluoride levels which remained in the purified water.
The resulting substance levels in the solar distilled lake water were close to the levels in boiled lake water, although testing was stopped abruptly. There was a slight increase in iron and fluoride levels in the solar still lake water and a decreased copper and total hardness levels.
“People don’t have to use these exact materials,” explained student Jonathan Goforth. “We just want the idea to be replicated. So, if you have reflective and transparent material, it’s possible to do.”
Effective Water Filtration is Possible using Inexpensive Resources
By Parrys Underwood, Shecoria Akins, Timothy Jones, Jordan Fluker, Josh Jenkins, Jalen Powell
The students planned to design a water filter that was easy to create and use as well as quick at removing harmful contaminants from dirty water. They decided sand filtration would be the most efficient method of manual filtration in communities that lack resources.
The students designed two different filtration systems. The first was a sand and rock filter system composed of gravel, sand, charcoal, a sponge, cheesecloth and plastic bottle. The second was composed with rice, a sponge, coffee filters, charcoal, a cheesecloth and plastic bottle. Based on experiments, the home reproduced water filter proved to be the best at removing contaminants from the water.
“We basically proved that a $2 filter could actually filter water more efficiently than the $70 filter you can buy in the store,” student Josh Jenkins said. “We turned black, opaque, muddy water into drinkable water. I drank it two weeks ago and I’m still alive right now. So, we proved that it works pretty well.”
Latest Headlines
-
02/12/2025
-
02/11/2025
-
02/10/2025
-
01/30/2025
-
12/03/2024