Welcome
 The Easley laboratory is focused on the development of novel
microanalytical techniques that allow us to perform unique experiments
on biological systems. We develop microfluidic methods and
small-volume hormone assays to study secretions from small numbers of
cells in the form of intact, primary tissue. Our current studies
are focused on primary adipose tissue and pancreatic islets extracted
from mice. Debilitating conditions such as diabetes, obesity, and
metabolic syndrome are fundamentally linked to these tissues.
The Easley laboratory is focused on the development of novel
microanalytical techniques that allow us to perform unique experiments
on biological systems. We develop microfluidic methods and
small-volume hormone assays to study secretions from small numbers of
cells in the form of intact, primary tissue. Our current studies
are focused on primary adipose tissue and pancreatic islets extracted
from mice. Debilitating conditions such as diabetes, obesity, and
metabolic syndrome are fundamentally linked to these tissues.
Specifically, our novel methods allow us to interrogate the dynamics of secretion of hormones such as insulin (from islets) and asprosin (from adipocytes) as well as secreted small molecules such as glycerol or intercellular messengers such as cyclic AMP. We are able to make these measurements on very small samples of fluid or tissue. In this light, it can be said that we develop small methods to measure small amounts of hormones from small biological samples. On the contrary, the results can be big, since we can extract information on the biological systems that is not possible with larger-scale, conventional methods.
Research in our laboratory spans several
scientific disciplines, such as fundamental analytical
chemistry, biochemistry, molecular and cellular biology, and device engineering. Please
browse through our website for more detailed discussions
of our research projects.
Group Members
PI | Grads | Undergrads | Postdocs | Former Members
GROUP PICTURE, Feb. 2026

Back row:
Eli Kelly,
Mainul Mazumder,
Josh Howell,
Jonathan Parris,
Sims McElroy, and
Hieu Pham.
Front row:
Prof. Easley,
Eloge Lwamba,
Carter Dees,
Darlington Nwankpa,
Alex Schamban,
Ryan Duerr, and
Sabita Dangol.
PRINCIPAL INVESTIGATOR (back to top)
 Christopher J. Easley, Ph.D. (email)
Christopher J. Easley, Ph.D. (email)
C. Dent Williams Professor, Bioanalytical Chemistry
and Associate Department Chair
(Dept. Webpage)
NIH Postdoctoral Fellow, Vanderbilt Medical Center (2006-08)
Ph.D. in Analytical Chemistry, University of Virginia (2002-06)
B.S.
in Chemistry, Mississippi State University (1998-2002)
GRADUATE STUDENTS (back to top)
 Sabita Dangol (email)
Sabita Dangol (email)
Ph.D. Candidate, Chemistry and Biochemistry (2022-present)

Josh Howell (email)
Ph.D. Candidate, Chemistry and Biochemistry (2023-present)

Eloge Lwamba (email)
Ph.D. Candidate, Chemistry and Biochemistry (2022-present)

Mainul Mazumder (email)
Ph.D. Candidate, Chemistry and Biochemistry (2020-present)

Darlington Nwankpa (email)
Ph.D. Candidate, Chemistry and Biochemistry (2024-present)

Jonathan Parris (email)
Ph.D. Candidate, Chemistry and Biochemistry (2025-present)

Hieu Pham (email)
Ph.D. Candidate, Chemistry and Biochemistry (2021-present)
UNDERGRADUATES (back to top)
 Carter Dees (email)
Carter Dees (email)
Biochemistry Major

Ryan Duerr (email)
Chemical Engineering Major

Eli Kelly (email)
Biochemistry Major

Sims McElroy (email)
Biomedical Sciences Major

Alex Schamban (email)
Chemistry Major
FORMER MEMBERS (back to top)
Former Graduate Students:
![]()
![]() Andresa B. Bezerra, Ph.D.; Assistant Teaching Professor at Elon U.
Andresa B. Bezerra, Ph.D.; Assistant Teaching Professor at Elon U.
![]()
![]() Jessica (Brooks) Schilling, Ph.D.; Product Dev. Engineer at Kerecis
Jessica (Brooks) Schilling, Ph.D.; Product Dev. Engineer at Kerecis
![]()
![]() Cheryl (DeJournette) Colquhoun, Ph.D.; Laboratory Manager at Auburn University
Cheryl (DeJournette) Colquhoun, Ph.D.; Laboratory Manager at Auburn University
![]()
![]() Kennon S. Deal, Ph.D.; Associate Professor at ABAC, Tifton, GA
Kennon S. Deal, Ph.D.; Associate Professor at ABAC, Tifton, GA
![]()
![]() Katarena Ford, Ph.D.; Senior Biologics Account Manager, Waters Corporation
Katarena Ford, Ph.D.; Senior Biologics Account Manager, Waters Corporation
![]()
![]() Leah A. Godwin, Ph.D.; Senior Chemist at CVI Engineering; Pensacola, FL
Leah A. Godwin, Ph.D.; Senior Chemist at CVI Engineering; Pensacola, FL
![]()
![]() Asanka Gurukandure, Ph.D.; Research Scientist at QPS Holdings; Newark, DE
Asanka Gurukandure, Ph.D.; Research Scientist at QPS Holdings; Newark, DE
![]()
![]() Tesfagebriel Hagos, M.S.; Medical Tech., DC Dept. of Forensic Sciences
Tesfagebriel Hagos, M.S.; Medical Tech., DC Dept. of Forensic Sciences
![]()
![]() Mark Holtan, Ph.D.; Research Instrument Coordinator at NC A&T University
Mark Holtan, Ph.D.; Research Instrument Coordinator at NC A&T University
![]()
![]() Jiaming Hu, Ph.D.; Professor at South China Normal Univ. (website)
Jiaming Hu, Ph.D.; Professor at South China Normal Univ. (website)
![]()
![]() Juan Hu, Ph.D.; Assistant Professor at San Diego St. U. (website)
Juan Hu, Ph.D.; Assistant Professor at San Diego St. U. (website)
![]()
![]() Yvette Kayirangwa, Ph.D.; Founder and CEO of Inkubito Wealth
Yvette Kayirangwa, Ph.D.; Founder and CEO of Inkubito Wealth
![]()
![]() Niamat Khuda, Ph.D.; Los Angeles Metro Area
Niamat Khuda, Ph.D.; Los Angeles Metro Area
![]()
![]() Amanda Kurian, Ph.D.; Research Scientist at QPS Holdings; Newark, DE
Amanda Kurian, Ph.D.; Research Scientist at QPS Holdings; Newark, DE
![]()
![]() Xiangpeng Li, Ph.D.; Assistant Professor at Florida State U. (website)
Xiangpeng Li, Ph.D.; Assistant Professor at Florida State U. (website)
![]()
![]() Md Mohibullah, Ph.D.; Scientist III at
Eurofins
Md Mohibullah, Ph.D.; Scientist III at
Eurofins
![]()
![]() Md Moniruzzman, Ph.D.; Postdoc at AU Chem. Eng.
Md Moniruzzman, Ph.D.; Postdoc at AU Chem. Eng.
![]()
![]() Jean Negou, Ph.D.; Engineer at Collier Venture, Inc.
Jean Negou, Ph.D.; Engineer at Collier Venture, Inc.
![]()
![]() Nan Shi, Ph.D.; Postdoc at UC Irvine
Nan Shi, Ph.D.; Postdoc at UC Irvine
![]()
![]() Joanne Seow, Ph.D.
Joanne Seow, Ph.D.
![]()
![]() Subramaniam (Mani) Somasundaram, Ph.D.; Senior Scientist II at Zimmer Biomet
Subramaniam (Mani) Somasundaram, Ph.D.; Senior Scientist II at Zimmer Biomet
Former Postdocs:
![]()
![]() William J. Ashby, Ph.D.; Sumo Robot League (Augusta, GA)
William J. Ashby, Ph.D.; Sumo Robot League (Augusta, GA)
![]()
![]() Adriana Avila, Ph.D.; Assistant Professor, Auburn Univ. (website)
Adriana Avila, Ph.D.; Assistant Professor, Auburn Univ. (website)
![]()
![]() Joonyul Kim, Ph.D.; CEO at Ciscovery Bio, Inc.
Joonyul Kim, Ph.D.; CEO at Ciscovery Bio, Inc.
![]()
![]() Subramaniam (Mani) Somasundaram, Ph.D.; Senior Scientist II at Zimmer Biomet
Subramaniam (Mani) Somasundaram, Ph.D.; Senior Scientist II at Zimmer Biomet
![]()
![]() Ajay Urgunde, Ph.D.; Visiting Assistant Professor at CSU Pueblo
Ajay Urgunde, Ph.D.; Visiting Assistant Professor at CSU Pueblo
Former Undergraduates:
Sawyer Stanley; Auburn University
MacKay Pfeiffer; UAB College of Medicine
Webber Alt; Auburn University
Patrick Rice; UAB College of Medicine
Jacob Sinopoli; Alabama College of Osteopathic Medicine
Madelyn James; Vanderbilt University, Graduate School
Max Blackwell; Auburn University
Joshua Baroody; Auburn University
Sidney Wasner; University of Huntsville, Graduate School
Haley Stephens; Auburn University
Swati Baskiyar; Auburn University
Joanne Seow (summer 2018); Auburn University, Ph.D. program
Dylan Holder; Emory University, Graduate School
Stuart Moon; University of Southern California, Graduate School
Andresa Bezerra; University of Sao Paulo, Brazil
Stephen Gass; Auburn University
Lauren Hoepfner; UAB School of Medicine, Birmingham, AL
Louis Jackson; UTenn School of Pharmacy, Memphis, TN
Zac Keenum; UAB School of Medicine, Birmingham, AL
Haley Medlen; UAB School of Medicine, Birmingham, AL
Jasmine Naik (REU 2013); Rowan University, Glassboro, NJ
Meagan E. Pilkerton, M.S.; Analytical Chemist at IEH Laboratories
Bailey Roberts; USF School of Medicine
Amanda Kelley Robertson; Blue Bell, PA
Ricky Scheuerle; USF School of Medicine, Tampa, FL
Rebecca S. Sollie, M.D.; U. of South Alabama School of Medicine, AL
Ferdous Torabinejad Finklea; Graduate Student, Dept. Chem. Eng., Auburn Univ.
Luke J. Vincent; UMC School of Medicine, Jackson, MS
Terrance Weeden, M.S.; Philadelphia College of Osteopathic Medicine
Research
Funding | Micro-Sampling | Biosensing | Droplets
Sampling secretions from small numbers of
endocrine cells
(back
to top)
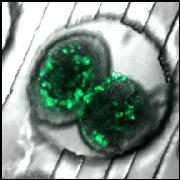
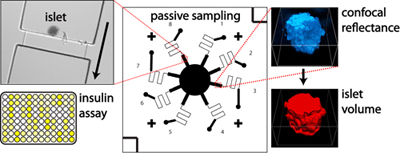 There
are several approaches that we utilize in order to sample secretions.
First, we have developed a passively-operated device for bulk secretion
sampling, in which the fluidic resistances of microfluidic channels
are exploited. The channel cross-sectional area and
length are carefully patterned to give specific fluidic resistances.
This way, murine pancreatic islets or adipocytes can be loaded upstream,
and flow rates for secretion sampling can be accurately controlled with
a handheld syringe by simply relying on the properties of the channels.
Bulk measurements of secretions from single islets can be made over
relatively long time periods (minutes to hours), and the method is
capable of high-throughput sampling from many islets, which is important
due to biological variability. Combining this sampling approach
with detection by insulin ELISA or our proximity assays (below) allows
us to collect the ensemble average of hundreds of islets while
preserving the single-islet data such as volume or architecture assayed
by confocal microscopy.
There
are several approaches that we utilize in order to sample secretions.
First, we have developed a passively-operated device for bulk secretion
sampling, in which the fluidic resistances of microfluidic channels
are exploited. The channel cross-sectional area and
length are carefully patterned to give specific fluidic resistances.
This way, murine pancreatic islets or adipocytes can be loaded upstream,
and flow rates for secretion sampling can be accurately controlled with
a handheld syringe by simply relying on the properties of the channels.
Bulk measurements of secretions from single islets can be made over
relatively long time periods (minutes to hours), and the method is
capable of high-throughput sampling from many islets, which is important
due to biological variability. Combining this sampling approach
with detection by insulin ELISA or our proximity assays (below) allows
us to collect the ensemble average of hundreds of islets while
preserving the single-islet data such as volume or architecture assayed
by confocal microscopy.
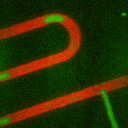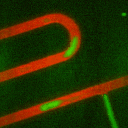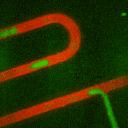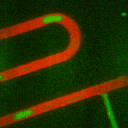 Secondly, we have developed methods using droplet microfliudics to
accurately measure temporal secretion profiles from single islets.
Picoliter volume aqueous droplets (green in video) are formed in in
silicone oil (red in video) downstream of islets during
glucose-stimulated insulin secretion. The microfluidic system is
ideal for minimizing dilution while preserving temporal information.
Droplets are shown to form with high monodispersity, at 0.470 nL volume
(<2% relative standard deviation, n = 3257 droplets). At high
temporal resolution, pulses of zinc secretion that match periods of
calcium oscillations were observed.
Secondly, we have developed methods using droplet microfliudics to
accurately measure temporal secretion profiles from single islets.
Picoliter volume aqueous droplets (green in video) are formed in in
silicone oil (red in video) downstream of islets during
glucose-stimulated insulin secretion. The microfluidic system is
ideal for minimizing dilution while preserving temporal information.
Droplets are shown to form with high monodispersity, at 0.470 nL volume
(<2% relative standard deviation, n = 3257 droplets). At high
temporal resolution, pulses of zinc secretion that match periods of
calcium oscillations were observed.
Cooperative sensing: Aptamers,
antibodies, and DNA 'circuits'
(back to top)
We are using cooperative approaches for detection
of a variety of protein and small-molecule targets.
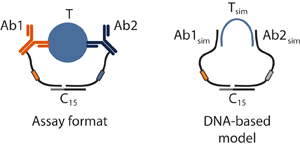 These approaches employ dual antibody-DNA conjugates or DNA aptamers to
serve as target-triggered signals in DNA-based circuits that employ
enzymatic ligation or fluorescence resonance energy transfer
(FRET). To gain a more fundamental understanding of these
types of assays, we have developed entirely DNA-based models to optimize
signal and background ligations. These models have allowed us to
significantly improve dynamic range, sensitivity, and limits of
detection for the assays. Additionally, we are developing an
electrochemical version of these assays, using square wave voltammetry
to detect the presence of a variety of protein targets. This
approach has excellent potential for point-of-care diagnostics.
Finally, we have explored options for aptamer selection using this
cooperative assembly mechanism. We have shown that it is possible
to use a known aptamer or antibody to find and enrich low abundance
aptamers saturated by a large pool of random-sequence DNA.
These approaches employ dual antibody-DNA conjugates or DNA aptamers to
serve as target-triggered signals in DNA-based circuits that employ
enzymatic ligation or fluorescence resonance energy transfer
(FRET). To gain a more fundamental understanding of these
types of assays, we have developed entirely DNA-based models to optimize
signal and background ligations. These models have allowed us to
significantly improve dynamic range, sensitivity, and limits of
detection for the assays. Additionally, we are developing an
electrochemical version of these assays, using square wave voltammetry
to detect the presence of a variety of protein targets. This
approach has excellent potential for point-of-care diagnostics.
Finally, we have explored options for aptamer selection using this
cooperative assembly mechanism. We have shown that it is possible
to use a known aptamer or antibody to find and enrich low abundance
aptamers saturated by a large pool of random-sequence DNA.
Droplet microfluidics
(back
to top)
The field of analytical chemistry has seen a
significant shift of effort toward droplet microfluidics over the past
five years, as researchers have begun to realize the enormous potential
of generating and controlling picoliter to nanoliter volume aqueous
droplets.
 These droplets can serve as ensembles of miniature chemical or
biochemical reactors, while also leveraging the inherent benefits of
analysis at the microfluidic scale. Our group has developed a secretion
sampling approach using droplet fluidics, and our recent work has
shown that aqueous droplets in oil can serve as a fluidic sample chopper
(mchopper). By enabling downstream
lock-in detection, this mchopper was shown to
reduce the detection limit of absorbance to the nanomolar range (500 nM
bromophenol blue), even over a very short 27-mm
optical path. The key functionality is the passive control of
droplet order, where the device design promotes consistent alternation
between sample and reference droplets (video shown here is in slow
motion; droplets generated at ~10 Hz from each channel in real time).
This technique is currently being applied to our cooperative FRET assays
for hormone secretion sampling.
These droplets can serve as ensembles of miniature chemical or
biochemical reactors, while also leveraging the inherent benefits of
analysis at the microfluidic scale. Our group has developed a secretion
sampling approach using droplet fluidics, and our recent work has
shown that aqueous droplets in oil can serve as a fluidic sample chopper
(mchopper). By enabling downstream
lock-in detection, this mchopper was shown to
reduce the detection limit of absorbance to the nanomolar range (500 nM
bromophenol blue), even over a very short 27-mm
optical path. The key functionality is the passive control of
droplet order, where the device design promotes consistent alternation
between sample and reference droplets (video shown here is in slow
motion; droplets generated at ~10 Hz from each channel in real time).
This technique is currently being applied to our cooperative FRET assays
for hormone secretion sampling.
Funding (back to top)
NSF, National Science Foundation (IIP-1549771, CBET-1403495, CBET-1067779, CBET-1337818, DUE-1102997)
NIH, National Institutes of Health (R35 GM162579 [2026 - present], R01 GM138828, R01 DK093810, R43 HG006078)
DHS S&T, Dept. of Homeland Security Science & Technology (DHS-70RSAT22CB0000002)
![]()

![]()

![]()
![]()
Pictures
Group Photo, May 2023
|
Group Photo, May 2021
|
Group Photo, Fall 2018
|
Group Photo, Summer 2017
|
Group Photo, Spring 2017
|
Group Photo, Summer 2016
|
Group Photo, Spring 2015
|
Group Photo, Spring 2013
|
Panorama Group Photo, Spring 2013
|
Group Photo, Spring 2012
|
Group Photo, Spring 2010
|
Pittcon 2010
|
Early days in the lab, 2009
|
Publications
Note: Full-text access requires journal subscription.
47. Gurukandure, A.; Mazumder, M. I.; Ortiz, K. G.; Kurian, A. S. N.; Somasundaram, S.; *Karimov, R. R.; *Easley, C. J., A Sensitive DNA Bowtie Sensor for Calibration-Free Sex Hormone Detection Using Analyte-DNA Conjugates and Multiple Redox Labels, ChemRxiv 2026, preprint published online. DOI: 10.26434/chemrxiv.15001151/v1
46. Kharal, S. P.; Mohibullah, M.; McDermott, T.; Easley, C. J.; *Louf, J. F., Confinement and hydraulic resistance appear to separately govern motility and path selection in Physarum polycephalum, J. R. Soc. Interface 2026, 23, 237, 20250873. https://doi.org/10.1098/rsif.2025.0873
45. Moniruzzaman, M.; Bezerra, A. B.; Mohibullah, M.; Judd, R. L.; Granneman, J. G.; Easley, C. J.*, Dynamic sampling from ex vivo adipose tissue using droplet-based microfluidics supports separate mechanisms for glycerol and fatty acid secretion, Lab Chip 2024, 24, 5020-5031. https://doi.org/10.1039/D4LC00664J
44. Kurian, A. S. N.; Mazumder, M. I.; Gurukandure, A.; Easley, C. J.*, An Electrochemical Proximity Assay (ECPA) for Antibody Detection Incorporating Flexible Spacers for Improved Performance, Anal. Bioanal. Chem. 2024, 416, 6529-6539. https://doi.org/10.1007/s00216-024-05546-9
43. Hu, J.; Easley, C. J.*, Development of a Mix-and-Read Assay for Human Asprosin using Antibody-Oligonucleotide Probes and Thermofluorimetric Analysis, Anal. Methods 2024, 16, 6057-6063. https://doi.org/10.1039/D3AY01175E
42. Gurukandure, A.; Somasundaram, S.; Kurian, A. S. N.; Khuda, N.; Easley, C. J.* Building a nucleic acid nanostructure with DNA-epitope conjugates for a versatile electrochemical protein detection platform, Anal. Chem. 2023, 95, 18122–18129. PDF
41. Kayirangwa, Y.; Mohibullah, M.; Easley, C. J.*, Droplet-based µChopper device with a 3D-printed pneumatic valving layer and a simple photometer for absorbance based fructosamine quantification in human serum, Analyst 2023, 148, 4810-4819. PDF
40. Kurian, A. S. N.; Gurukandure, A.; Dovgan, I.; Kolodych, S.; Easley, C. J.*, Thermofluorimetric Analysis (TFA) using Probes with Flexible Spacers: Application to Direct Antibody Sensing and to Antibody-Oligonucleotide (AbO) Conjugate Valency Monitoring, Anal. Chem. 2023, 95, 11680–11686. PDF
39. Khuda, N.; Somasundaram, S.; Urgunde, A.; Easley, C. J.*, Ionic Strength and Hybridization Position Near Gold Electrodes Can Significantly Improve Kinetics in DNA-Based Electrochemical Sensors, ACS Appl. Mater. Interfaces 2023, 15, 5019-5027. PDF
38. Khuda, N.; Somasundaram, S.; Easley, C. J.*, Electrochemical Sensing of the Peptide Drug Exendin-4 using a Versatile Nucleic Acid Nanostructure, ACS Sens. 2022, 7, 784-789. PDF
37. Shi, N.; Mohibullah, M.; Easley, C. J.*, Active Flow Control and Dynamic Analysis in Droplet Microfluidics, Annu. Rev. Anal. Chem. 2021, 14, 133-153. PDF
36. Bezerra, A. B.; Kurian, A. S. N.; Easley, C. J.*, Nucleic-Acid Driven Cooperative
Bioassays using Probe Proximity or Split-Probe Techniques,
Anal. Chem. 2021, 93, 198-214.
PDF
- Invited for 2021 Special Issue: Fundamental and Applied
Reviews in Analytical Chemistry
35. Shi, N.; Easley, C. J.*, Programmable µChopper Device with On-Chip
Droplet Mergers for Continuous Assay Calibration,
Micromachines 2020, 11, 620. PDF
- Invited contribution to special issue on "Droplet Microfluidics"
34. Shi, N.; Moniruzzaman, M.; Easley, C. J.*, “Tissue Engineering and Analysis in Droplet Microfluidics,” in Droplet Microfluidics; Ren, C., Lee, A., Eds.; Royal Society of Chemistry (Cambridge, UK) 2020, in press.
33. Hu, J.; Li, X.; Judd, R. L.; Easley, C. J.*, Rapid lipolytic oscillations in ex-vivo adipose tissue explants revealed through microfluidic droplet sampling at high temporal resolution, Lab Chip 2020, 20, 1503-1512. PDF
32. Holtan, M. D.; Somasundaram, S.; Khuda, N.; Easley, C. J.*, Non-Faradaic Current Suppression in DNA Based Electrochemical Assays with a Differential Potentiostat Anal. Chem. 2019, 91, 15833-15839. PDF
31. Somasundaram, S.; *Easley, C. J., A Nucleic Acid Nanostructure Built through On-electrode Ligation for Electrochemical Detection of a Broad Range of Analytes, J. Am. Chem. Soc. 2019, 141, 11721-11726. PDF
30. Li, X.; Hu, J.; Easley, C. J.*, Automated microfluidic droplet sampling with integrated, mix-and-read immunoassays
to resolve endocrine tissue secretion dynamics,
Lab Chip 2018, 18, 2926-2935. PDF
- Selected as Cover Article (inside front)
- Featured in the journal's top 10% list, "Recent Hot Articles" (link)
29. Negou, J. T.; Hu, J.; Li, X.; Easley, C. J.*, Advancement of analytical modes in a multichannel, microfluidic droplet-based sample chopper employing phase-locked detection,
Anal. Methods 2018, 10, 3436-3443.
PDF
- Selected as Cover Article
28. Easley, C. J.; Regan, F.; Roper, M. G.; Martin, R. S.*, In celebration of the 60th birthday of
2 microfluidics pioneers: Professor Susan Lunte and Professor James Landers,
Anal. Methods 2018, 10, 3433-3435.
PDF
- Editorial
27. Easley, C. J.*, Eric Lagally (Ed.): Microfluidics and nanotechnology: biosensing to the
single molecule limit,
Anal. Bioanal. Chem. 2018, in press, DOI: 10.1007/s00216-018-1193-5.
PDF
- Book review
26. Easley, C. J.; Kim, J.; Hu, J.; Holtan, M. D.; Somasundaram, S.; Shannon, C. G., Thermally Resolved Molecule Assays, U.S. Patent 9,995,680; June 12, 2018.
25. Somasundaram, S.; Holtan, M. D.; Easley, C. J.*, Understanding signal and background in a thermally resolved, single-branched DNA assay using square wave voltammetry, Anal. Chem. 2018, 90, 3584-3591. PDF
24. Li, X.; Easley, C. J.*, Microfluidic systems for studying dynamic function of adipocytes and adipose tissue, Anal. Bioanal. Chem.
2018, 410, 791-800. PDF
- Critical Review; Invited submission to the ABC 16th Anniversary Issue
23. Hu, J.; Easley, C. J.*, Homogeneous Assays of Second Messenger Signaling and Hormone Secretion using Thermofluorimetric Methods that Minimize Calibration Burden, Anal. Chem. 2017, 89, 8517–8523. PDF
22. Negou, J. T.; Avila, L. A.; Li, X.; Hagos, T. M.; Easley, C. J.*, An automated microfluidic droplet-based sample chopper for detection of small fluorescence differences using lock-in analysis, Anal. Chem. 2017, 89, 6153-6159. PDF
21. Li, X.; Brooks, J. C.; Hu, J.; Ford, K. I.; Easley, C. J.*, 3D-templated, fully automated microfluidic input/output multiplexer for endocrine tissue culture and secretion sampling, Lab Chip 2017, 17, 341-349. PDF
20. Brooks, J. C.; Judd, R. L.; Easley, C. J.*, Culture and Sampling of Primary Adipose Tissue in Practical Microfluidic Systems, in Methods in Molecular Biology: Thermogenic Fat - Methods and Protocols, Humana Press/Springer (New York) 2017, in press, DOI: 10.1007/978-1-4939-6820-6_18
19. Brooks, J. C.; Ford, K. I.; Holder, D. H.; Holtan, M. D.; Easley, C. J.*, Macro-to-micro interfacing to microfluidic channels using 3D-printed templates: Application to time-resolved secretion sampling of endocrine tissue, Analyst 2016, 141, 5714-5721. PDF
18. Kerscher, P.; Turnbull, I. C.; Hodge, A. J.; Kim, J.; Seliktar, D.; Easley, C. J.; Costa, K. D.; Lipke, E.*, Direct Hydrogel Encapsulation of Pluripotent Stem Cells Enables Ontomimetic Differentiation and Growth of Engineered Human Heart Tissues, Biomaterials 2016, 83, 383-395. PDF
17. Hu, J.; Wang, T.; Shannon, C.; Easley, C. J. Electrochemical Proximity Assay. US Patent 9,335,292 B2, May 10, 2016.
16. Kim, J.; Hu, J.; Bezerra, A. B.; Holtan, M. D.; Brooks, J. C.; Easley, C. J.*, Protein Quantification using Controlled DNA Melting Transitions in Bivalent Probe Assemblies, Anal. Chem. 2015, 87, 9576-9579. PDF
15. Juan Hu, Joonyul Kim, and Christopher J. Easley*,
Quantifying Aptamer-Protein Binding via Thermofluorimetric Analysis, Anal. Methods
2015, 7, 7358-7362. PDF
- Dr. Easley featured in "Emerging Investigators" issue (link)
14. Leah A. Godwin, Jessica C. Brooks, Lauren D. Hoepfner, Desiree Wanders, Robert L. Judd, and Christopher J. Easley*,
A Microfluidic Interface Design for the Culture and Sampling of Adiponectin from Primary Adipocytes, Analyst
2015, 140, 1019-1025. PDF
- Selected as Cover Article
- Featured as Analyst Hot Article
13. Branson A. Maynard, Jessica C. Brooks, Emily E. Hardy, Christopher J. Easley, and Anne E. V. Gorden*, Synthesis, structural characterization, electronic spectroscopy, and microfluidic detection of Cu+2 and UO2+2 [di-tert-butyl-salphenazine] complexes, Dalton Trans. 2015, 44, 4428-4430. PDF
12. Jiaming Hu, Yajiao Yu, Jessica C. Brooks, Leah A. Godwin, Subramaniam Somasundaram, Ferdous Torabinejad, Joonyul Kim, Curtis Shannon*, and Christopher J. Easley*, A Reusable Electrochemical Proximity Assay for Highly Selective, Real-Time Protein Quantitation in Biological Matrices, J. Am. Chem. Soc. 2014, 136, 8467-8474. PDF
11. Landers, J. P.; Bienvenue, J. M.; Legendre, L. A.; Easley, C. J.; Karlinsey, J. M., Integrated microfluidic analysis systems, US Patent 8,916,375 B2, Dec. 23, 2014
10. Cheryl J. DeJournette, Joonyul Kim, Haley Medlen, Xiangpeng Li, Luke J. Vincent, and Christopher J. Easley, Creating Biocompatible Oil-Water Interfaces without Synthesis: Direct Interactions between Primary Amines and Carboxylated Perfluorocarbon Surfactants, Anal. Chem. 2013, 85, 10556-10564. PDF
9. Leah A. Godwin, Kennon S. Deal, Lauren D. Hoepfner, Louis A. Jackson, Christopher J. Easley, Measurement of Microchannel Fluidic Resistance with a Standard Voltage Meter, Anal. Chim. Acta 2013, 758, 101-107. PDF
8. Jiaming Hu, Tanyu Wang, Joonyul Kim, Curtis Shannon*, Christopher J. Easley*, Quantitation of femtomolar protein levels via direct readout with the electrochemical proximity assay, J. Am. Chem. Soc. 2012, 134, 7066–7072. PDF
7. Daniel W. Horn; K. P. Tracy; Christopher J. Easley, Virginia A. Davis, Lysozyme Dispersed Single-Walled Carbon Nanotubes: Interaction and Activity, J. Phys. Chem. C 2012, 116, 10341–10348. PDF
6. Kennon S. Deal and Christopher J. Easley, A Self-Regulated, Droplet-Based Sample Chopper for Microfluidic Absorbance Detection, Analytical Chemistry 2012, 84, 1510–1516. PDF
5. Easley, C. J.; Karlinsey, J. M.; Leslie, D. C.; Begley, M. R.; Landers, J. P. Passive components for micro-fluidic flow profile shaping and related method thereof. US Patent 8,220,493 B2, July 17, 2012
4. Leah A. Godwin, Meagan E. Pilkerton, Kennon S. Deal, Desiree Wanders, Robert L. Judd, and Christopher J. Easley, A passively operated microfluidic device for stimulation and secretion sampling of single pancreatic islets, Analytical Chemistry, 83 (2011) 7166–7172. PDF
3. Jiaming Hu and Christopher J. Easley, A Simple and Rapid Approach for Measurement of Dissociation Constants of DNA Aptamers against Proteins and Small Molecules via Automated Microchip Electrophoresis, Analyst, 136 (2011) 3461-3468. PDF
2. Joonyul Kim and Christopher J. Easley, Isothermal DNA Amplification in Bioanalysis: Strategies and Applications, Bioanalysis, 3 (2011) 227-239. Author's PDF, Publisher's PDF
1. Joonyul Kim, Jiaming Hu, Rebecca S. Sollie, Christopher J. Easley, Improvement of sensitivity and dynamic range in proximity ligation assays by asymmetric connector hybridization, Analytical Chemistry, 82 (2010) 6976-6982. PDF
Prior to Auburn University (back to top)
13. Christopher J. Easley, Jonathan V. Rocheleau, W. Steven Head, and
David W. Piston, Quantitative measurement of zinc secretion from
pancreatic islets with high temporal resolution using droplet-based
microfluidics,
Analytical Chemistry, 81 (2009) 9086-9095.
PDF
- Full-page
Research Profile by
Nancy Lamontagne
12. Daniel C. Leslie, Christopher J. Easley, Erkin Seker, James M. Karlinsey,
Marcel Utz, Matthew R. Begley, and James P. Landers, Frequency-specific flow
control in microfluidic circuits with passive elastomeric features,
Nature Physics, 5 (2009) 231-235.
PDF
- Editors' Choice, Science, 23 (2009) 1539.
PDF
- Research Highlight, Lab on a Chip, 9 (2009)
861.
PDF
11. Christopher J. Easley, Richard K. P. Benninger, Jesse H. Shaver, W.
Steven Head, and David W. Piston, Rapid and inexpensive fabrication of polymeric
microfluidic devices via toner transfer masking,
Lab on a Chip, 9 (2009) 1119-1127.
PDF
- Research Highlight, Nature Methods, 6 (2009)
194.
PDF
10. Shu Mao, Richard K. P. Benninger, Yuling Yan, Chutima Petchprayoon, David Jackson, Christopher J. Easley, David W. Piston, and Gerard Marriott, Optical lock-in detection of fluorescence resonance energy transfer using synthetic and genetically-encoded optical switches, Biophysical Journal, 94 (2008) 4515-4524. PDF
9. Christopher J. Easley, Joseph A. C. Humphrey, and James P. Landers, Thermal isolation of microchip reaction chambers for rapid non-contact DNA amplification, Journal of Micromechanics and Microengineering, 17 (2007) 1758-1766. PDF
8. Ki-Ho Han, Rachel D. McConnell, Christopher J. Easley, Joan M. Bienvenue, Jerome P. Ferrance, James P. Landers, and A. Bruno Frazier, An active microfluidic system packaging technology, Sensors and Actuators B: Chemical, 122 (2007) 337-346. PDF
7. Christopher J. Easley, James M. Karlinsey, Joan M. Bienvenue, Lindsay A. Legendre, Michael G. Roper, Sanford H. Feldman, Molly A. Hughes, Erik L. Hewlett, Tod J. Merkel, Jerome P. Ferrance, and James P. Landers, A fully-integrated microfluidic genetic analysis system with sample in-answer out capability, Proceedings of the National Academy of Sciences USA, 103 (2006) 19272-19277. PDF [Highlighted in Science as an Editor's Choice, 19 January: 315 (2007) 5810. PDF ; also in Nature Biotechnology as a Research Highlight, January: 25 (2007) 69; and in Analytical Chemistry as a Bio Sphere news article, February: 79 (2007) 809.]
6. Michael G. Roper, Christopher J. Easley, Lindsay A. Legendre, Joseph A. C. Humphrey, and James P. Landers, Infrared temperature control system for a completely noncontact polymerase chain reaction in microfluidic chips, Analytical Chemistry, 79 (2007) 1294-1300. PDF
5. Weidong Cao, Christopher J. Easley, Jerome P. Ferrance, and James P. Landers, Chitosan as a polymer for pH-induced DNA capture in a totally aqueous system, Analytical Chemistry, 78 (2006) 7222-7228. PDF
4. Christopher J. Easley, James M. Karlinsey, and James P. Landers, On-chip pressure injection for integration of infrared-mediated DNA amplification with electrophoretic separation, Lab on a Chip, 6 (2006) 601-610. [Cover Article] PDF
3. Guihua Eileen Yue, Michael G. Roper, Erin D. Jeffery, Christopher J. Easley, Catherine Balchunas, James P. Landers, and Jerome P. Ferrance, Glass microfluidic devices with thin membrane voltage junctions for electrospray mass spectrometry, Lab on a Chip, 5 (2005) 619-627. PDF
2. Christopher J. Easley, Lindsay A. Legendre, Michael G. Roper, and James P. Landers, Extrinsic Fabry-Perot interferometry for non-contact temperature control of nanoliter volume enzymatic reactions in glass microchips, Analytical Chemistry, 77 (2005) 1038-1045. PDF
1. Christopher J. Easley, Lian Ji Jin, Katja B. Presto Elgstoen, Egil Jellum, James P. Landers, and Jerome P. Ferrance, Capillary electrophoresis with laser-induced fluorescence detection for laboratory diagnosis of galactosemia, Journal of Chromatography A, 1004 (2003) 29-37. PDF
News and Tweets
Tweets by EasleyLabAugust 2018: Two more articles selected for journal covers!

 Two recent manuscripts from the Easley group were featured on journal covers in recent issues of the Royal Society of Chemistry’s journals, Analytical Methods and Lab on a Chip.
The first authors of the articles were Jean Negou and Xiangpeng Li, and both of the manuscripts reported
our recent developments in microfluidic flow control using valves to form, manipulate, and analyte aqueous-in-oil droplets.
Jean's work showed that multiple analytical modes are available with valve-controlled droplets, while Li's work showed that
droplets can be used to provide high temporal resolution sampling of hormone secretion and quantification with
in-droplet homogeneous protein assays. Congratulations to Jean, Li, and all of the co-authors!
Two recent manuscripts from the Easley group were featured on journal covers in recent issues of the Royal Society of Chemistry’s journals, Analytical Methods and Lab on a Chip.
The first authors of the articles were Jean Negou and Xiangpeng Li, and both of the manuscripts reported
our recent developments in microfluidic flow control using valves to form, manipulate, and analyte aqueous-in-oil droplets.
Jean's work showed that multiple analytical modes are available with valve-controlled droplets, while Li's work showed that
droplets can be used to provide high temporal resolution sampling of hormone secretion and quantification with
in-droplet homogeneous protein assays. Congratulations to Jean, Li, and all of the co-authors!
December 2018: Three Ph.D. graduates
 Congratulations again to Dr. Juan Hu, Dr. Katarena Ford, and Dr. Mark Holtan!
We wish you all the best, although we know you have bright futures ahead of you.
This year was particularly special for Prof. Easley, since he was able to carry out
the hooding ceremony with three students during the same graduation event.
Congratulations again to Dr. Juan Hu, Dr. Katarena Ford, and Dr. Mark Holtan!
We wish you all the best, although we know you have bright futures ahead of you.
This year was particularly special for Prof. Easley, since he was able to carry out
the hooding ceremony with three students during the same graduation event.
December 2018: Technologies from Easley and Shannon Labs licensed by Innamed, Inc.
 Innamed, Inc. (website) has chosen to license two technologies
developed by both Prof. Easley's and Prof. Shannon's laboratories to help with their at-home and
point-of-care blood testing devices. Our technology should help Innamed as they move toward their
goals of improving personalized medicine and at-home testing, and we look forward to a strong
collaborative relationship with the company. Thanks go to Troy Brady and the Auburn IAC
(website) for all of their invaluable help
along the way.
This photo was taken during a technology training session held in the Easley lab in December of 2017.
Innamed, Inc. (website) has chosen to license two technologies
developed by both Prof. Easley's and Prof. Shannon's laboratories to help with their at-home and
point-of-care blood testing devices. Our technology should help Innamed as they move toward their
goals of improving personalized medicine and at-home testing, and we look forward to a strong
collaborative relationship with the company. Thanks go to Troy Brady and the Auburn IAC
(website) for all of their invaluable help
along the way.
This photo was taken during a technology training session held in the Easley lab in December of 2017.
November 2018: Mark successfully defended his Ph.D. work!
 Mark impressed his committee during his Ph.D. defense by presenting his work entitled
"Passive Microfluidic Flow Control Components and a Custom Differential Potentiostat."
Dr. Holtan will soon move to the Durham, NC area. Our lab
(and the entire department) will surely feel your absence, particularly when instrumentation
needs some repair work. What is your consulting fee again?
Anyway, congratulations, Dr. Holtan!
Mark impressed his committee during his Ph.D. defense by presenting his work entitled
"Passive Microfluidic Flow Control Components and a Custom Differential Potentiostat."
Dr. Holtan will soon move to the Durham, NC area. Our lab
(and the entire department) will surely feel your absence, particularly when instrumentation
needs some repair work. What is your consulting fee again?
Anyway, congratulations, Dr. Holtan!
September 2018: Kat successfully defended her Ph.D. work!
 Katarena (Kat) Ford passed her Ph.D. defense by presenting her work entitled
"The Development of a Nucleic Acid-Directed Switch and Proximity Dependent Protein
Assays using Electrochemical Techniques." Dr. Ford will
soon move to Nashville, TN to work as a postdoctoral fellow in Rena Robinson's
lab at Vanderbilt University. Congratulations, Kat! We have some work ahead of us to try
to fill your shoes in the lab.
Katarena (Kat) Ford passed her Ph.D. defense by presenting her work entitled
"The Development of a Nucleic Acid-Directed Switch and Proximity Dependent Protein
Assays using Electrochemical Techniques." Dr. Ford will
soon move to Nashville, TN to work as a postdoctoral fellow in Rena Robinson's
lab at Vanderbilt University. Congratulations, Kat! We have some work ahead of us to try
to fill your shoes in the lab.
July 2018: Juan successfully defended her Ph.D. work!
 Juan Hu succesfully defended her Ph.D. work entitled
"Development of thermally regulated homogeneous assays and droplet-based microfluidic
tools for probing secretion dynamics from pancreatic and adipose tissues." Dr. Hu will
soon move on to the Scripps Research Institute in Jupiter, FL to work as a postdoctoral fellow
in Brian Paegel's lab. Congratulations, Juan! We will all miss you!
Juan Hu succesfully defended her Ph.D. work entitled
"Development of thermally regulated homogeneous assays and droplet-based microfluidic
tools for probing secretion dynamics from pancreatic and adipose tissues." Dr. Hu will
soon move on to the Scripps Research Institute in Jupiter, FL to work as a postdoctoral fellow
in Brian Paegel's lab. Congratulations, Juan! We will all miss you!
March 27, 2018: Juan Hu selected for Dean's Research Award
 Contratulations to Juan Hu, who was selected for the prestigious COSAM Dean's Research Award
for doctoral candidates! She is the first student from the Easley laboratory to receive this award.
Clearly, the work that Juan has accomplished toward her Ph.D. has been of the highest quality. We
are all very proud of her accomplishments and her award!
Contratulations to Juan Hu, who was selected for the prestigious COSAM Dean's Research Award
for doctoral candidates! She is the first student from the Easley laboratory to receive this award.
Clearly, the work that Juan has accomplished toward her Ph.D. has been of the highest quality. We
are all very proud of her accomplishments and her award!
December 2017: Mani successfully defended his Ph.D. work!
 Subramaniam (Mani) Somasundaram passed his Ph.D. defense by presenting his work entitled
"Development of Oligonucleotide, Small Molecule, and Protein Assays using Square-wave
Voltammetry with Electrode-bound Nucleic Acid Nanostructures." This makes three successful
Ph.D. defenses from our group in 2017. Congratulations, Mani! Or perhaps we should say:
Congratulations, Dr. Mani!
Subramaniam (Mani) Somasundaram passed his Ph.D. defense by presenting his work entitled
"Development of Oligonucleotide, Small Molecule, and Protein Assays using Square-wave
Voltammetry with Electrode-bound Nucleic Acid Nanostructures." This makes three successful
Ph.D. defenses from our group in 2017. Congratulations, Mani! Or perhaps we should say:
Congratulations, Dr. Mani!
November 2017: Jean successfully defended his Ph.D. work!
 Jean Negou passed his Ph.D. defense by presenting his work entitled
"Microfluidic Circuit Designs for Nanoliter-Scale Flow Control and Highly Sensitive
Quantitative Bioanalysis." Here is a photo from the December graduation just after
the hooding ceremony. Congratulations, Jean! You will surely be missed!
Jean Negou passed his Ph.D. defense by presenting his work entitled
"Microfluidic Circuit Designs for Nanoliter-Scale Flow Control and Highly Sensitive
Quantitative Bioanalysis." Here is a photo from the December graduation just after
the hooding ceremony. Congratulations, Jean! You will surely be missed!
July 2017: Li successfully defended his Ph.D. work!
 Xiangpeng Li sailed through his Ph.D. defense by presenting his work entitled
"Automated microfluidic device development for metabolism, nutrient uptake,
and hormone secretion analyses of primary endocrine tissues." Dr. Li will
soon move to San Fransisco, CA to work as a postdoctoral fellow in Adam Abate's
lab at UCSF. Congratulations, Li! We will certainly miss you in the Easley lab.
Xiangpeng Li sailed through his Ph.D. defense by presenting his work entitled
"Automated microfluidic device development for metabolism, nutrient uptake,
and hormone secretion analyses of primary endocrine tissues." Dr. Li will
soon move to San Fransisco, CA to work as a postdoctoral fellow in Adam Abate's
lab at UCSF. Congratulations, Li! We will certainly miss you in the Easley lab.
Teaching
Undergraduate Courses
CHEM 1040 - Fundamental Chemistry II
CHEM 3050 - Analytical Chemistry (Quantitative Analysis)
CHEM 4130 - Instrumental Analysis
(Java Applet files)
Graduate Courses
CHEM 7500 - Advanced Analytical Chemistry
CHEM 7530 - Advances in Bioanalytical Chemistry
Outreach Activities
Module development and teacher training for Math and Science
Partnership (MSP) with Tuskegee University
(AU-MSP website,
NanoBio website).
Award: DUE-1102997

![]()
Links of Interest
Auburn Chemistry and Biochemistry
Auburn Hardware Discounts
Diabetes Related:
Boshell
Diabetes and Metabolic Diseases Research Program
UAB
Comprehensive Diabetes Center
Midwest Islet Club
Power Supplies:
Trek, Inc.
Microscopy:
Molecular
Expressions
Oligonucleotide Synthesis:
Integrated DNA Technologies
NUPACK by DNA Group
at CalTech
Chemistry:
Kd Calculations
DNA melting
temperature calculations
Properties of PDMS
Biology:
Beta Cell Biology
Consortium
Pancreatic Islet Secretions
Insulin and Glucagon Secretion
Mouse Surgeries
Microchip Fabrication:
BYU Cleanroom
Image Processing:
Learning Resources
ImageJ (image
analysis shareware from NIH)
Reading TIFFs
with Matlab
Other:
All About
Circuits
MedBio
Aptamer
Database
Aptamer
Base
Lucigen
 Back row (from left): Hieu Pham, Eloge Lwamba, MacKay Pfeiffer, Andresa Bezerra, Prof. Easley,
Josh Howell, Yvette Kayirangwa, Asanka Gurukandure, Joanne Seow, Sabita Dangol, Ajay Urgunde, and Md Mohibullah.
Back row (from left): Hieu Pham, Eloge Lwamba, MacKay Pfeiffer, Andresa Bezerra, Prof. Easley,
Josh Howell, Yvette Kayirangwa, Asanka Gurukandure, Joanne Seow, Sabita Dangol, Ajay Urgunde, and Md Mohibullah. Back row (from left): Andresa Bezerra, Amanda Kurian, Joanne Seow, Md Moniruzzaman, Jacob Sinopoli,
Md Mohibullah, and Yvette Kayirangwa.
Back row (from left): Andresa Bezerra, Amanda Kurian, Joanne Seow, Md Moniruzzaman, Jacob Sinopoli,
Md Mohibullah, and Yvette Kayirangwa. Back row (from left): Nan Shi, Md Mohibullah, Andresa Bezerra, Swati Baskiyar, Katarena Ford,
Asanka Gurukandure Gedara, Mani Somasundaram, and Mark Holtan.
Back row (from left): Nan Shi, Md Mohibullah, Andresa Bezerra, Swati Baskiyar, Katarena Ford,
Asanka Gurukandure Gedara, Mani Somasundaram, and Mark Holtan. Back row (from left): Adriana Avila, Mani Somasundaram, Mark Holtan, and Jean Negou.
Back row (from left): Adriana Avila, Mani Somasundaram, Mark Holtan, and Jean Negou.
 Back row (from left): Joonyul Kim, Mark Holtan, Mani Somasundaram, and Adriana Avila.
Back row (from left): Joonyul Kim, Mark Holtan, Mani Somasundaram, and Adriana Avila. Back row (from left): Xiangpeng Li, Chris Easley, Joonyul Kim, Katarena Ford, Dylan Holder, Mani Somasundaram, and Stephen Gass.
Back row (from left): Xiangpeng Li, Chris Easley, Joonyul Kim, Katarena Ford, Dylan Holder, Mani Somasundaram, and Stephen Gass. Back row (from left): Ricky Scheuerle, Xiangpeng Li, Jiaming Hu, Ferdous Finklea, Haley Medlen, Lauren Hoepfner, Leah Godwin, Jess Brooks, Will Ashby, and Zac Keenum.
Back row (from left): Ricky Scheuerle, Xiangpeng Li, Jiaming Hu, Ferdous Finklea, Haley Medlen, Lauren Hoepfner, Leah Godwin, Jess Brooks, Will Ashby, and Zac Keenum. Group size was doubled using an iPhone app!
Group size was doubled using an iPhone app!
 Back row (from left): Joonyul Kim, Louis Jackson, and Lauren Hoepfner.
Back row (from left): Joonyul Kim, Louis Jackson, and Lauren Hoepfner. Pictured (from left): Leah Godwin, Zac Keenum, Meagan Pilkerton, Jiaming Hu, Kennon Deal,
Chris Easley, Rebecca Sollie, and Cheryl DeJournette.
Pictured (from left): Leah Godwin, Zac Keenum, Meagan Pilkerton, Jiaming Hu, Kennon Deal,
Chris Easley, Rebecca Sollie, and Cheryl DeJournette.
 Group Dinner at Pittcon 2010, Orlando, FL
Group Dinner at Pittcon 2010, Orlando, FL
 Early days in the lab with Jiaming, Dr. Easley, and Leah.
Early days in the lab with Jiaming, Dr. Easley, and Leah.