Tridymite and Cristobalite - SiO2
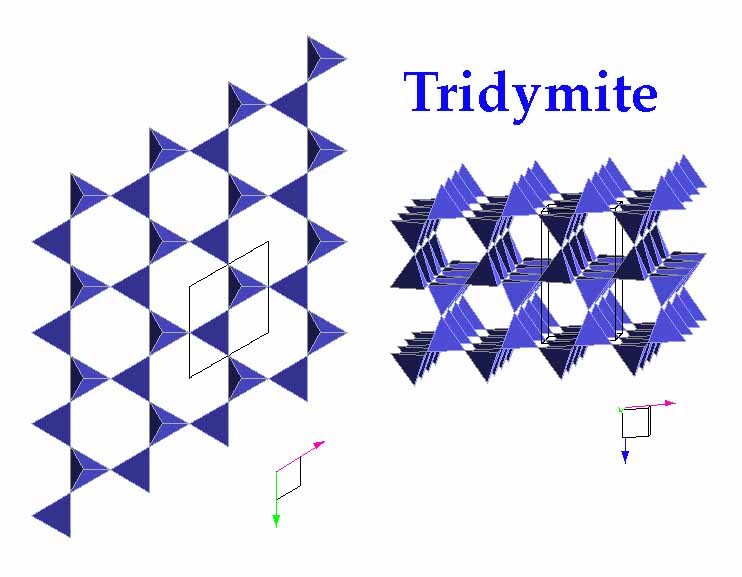 Tridymite
has (SiO4)-4 tetrahedra arranged in sheets, and the
tetrahedra alternately point up or down to share oxygen with the tetrahedra
of other sheets. Note that although this arrangement might seem similar
to (SiO4)-4 tetrahedra in the cyclosilicates, in
tridymite each six-sided ring is linked to those of the sheet above andf
below (in cyclosilicates, each tetrahedra of a given ring 'points' in the
same direction. Tridymite is common in certain siliceous volcanic rocks.
Tridymite
has (SiO4)-4 tetrahedra arranged in sheets, and the
tetrahedra alternately point up or down to share oxygen with the tetrahedra
of other sheets. Note that although this arrangement might seem similar
to (SiO4)-4 tetrahedra in the cyclosilicates, in
tridymite each six-sided ring is linked to those of the sheet above andf
below (in cyclosilicates, each tetrahedra of a given ring 'points' in the
same direction. Tridymite is common in certain siliceous volcanic rocks.
 Cristobalite
has an isometric structure, in which six-membered rings of (SiO4)-4
tetrahedra are stacked parallel to (111). The view in the accompanying
photo is along [111], and each tetrahedron occurs at the corner or face
of a regular cube (the unit cell). This arrangement of tetrahedra is analogous
to the stacking of spheres in cubic closest packing. Note the increase
in symmetry, from hexagonal to isometric, with increasing temperature in
the silica polymorphs.
Cristobalite
has an isometric structure, in which six-membered rings of (SiO4)-4
tetrahedra are stacked parallel to (111). The view in the accompanying
photo is along [111], and each tetrahedron occurs at the corner or face
of a regular cube (the unit cell). This arrangement of tetrahedra is analogous
to the stacking of spheres in cubic closest packing. Note the increase
in symmetry, from hexagonal to isometric, with increasing temperature in
the silica polymorphs.
 Tridymite
has (SiO4)-4 tetrahedra arranged in sheets, and the
tetrahedra alternately point up or down to share oxygen with the tetrahedra
of other sheets. Note that although this arrangement might seem similar
to (SiO4)-4 tetrahedra in the cyclosilicates, in
tridymite each six-sided ring is linked to those of the sheet above andf
below (in cyclosilicates, each tetrahedra of a given ring 'points' in the
same direction. Tridymite is common in certain siliceous volcanic rocks.
Tridymite
has (SiO4)-4 tetrahedra arranged in sheets, and the
tetrahedra alternately point up or down to share oxygen with the tetrahedra
of other sheets. Note that although this arrangement might seem similar
to (SiO4)-4 tetrahedra in the cyclosilicates, in
tridymite each six-sided ring is linked to those of the sheet above andf
below (in cyclosilicates, each tetrahedra of a given ring 'points' in the
same direction. Tridymite is common in certain siliceous volcanic rocks.
 Cristobalite
has an isometric structure, in which six-membered rings of (SiO4)-4
tetrahedra are stacked parallel to (111). The view in the accompanying
photo is along [111], and each tetrahedron occurs at the corner or face
of a regular cube (the unit cell). This arrangement of tetrahedra is analogous
to the stacking of spheres in cubic closest packing. Note the increase
in symmetry, from hexagonal to isometric, with increasing temperature in
the silica polymorphs.
Cristobalite
has an isometric structure, in which six-membered rings of (SiO4)-4
tetrahedra are stacked parallel to (111). The view in the accompanying
photo is along [111], and each tetrahedron occurs at the corner or face
of a regular cube (the unit cell). This arrangement of tetrahedra is analogous
to the stacking of spheres in cubic closest packing. Note the increase
in symmetry, from hexagonal to isometric, with increasing temperature in
the silica polymorphs.